India’s Electric Vehicle Market: A Look Back at 2023 Get...
Read MoreVineet Dravid
Founder, CEO oorja
Solving the Thermal Runaway Conundrum in EV Batteries
According to a survey by CarTrade Tech on barriers to two wheeler EV adoption in India, 45% of respondents said newspaper articles on EVs catching fire was the biggest deterrent to them buying an EV. This isn’t surprising given that thermal runaway(TR) is currently the most critical safety challenge in the adoption of EVs, especially the case in air cooled vehicles: mainly electric two and three wheelers.
Unfortunately, thermal runaway is a very poorly understood topic both by technologists and the general population alike for various reasons. A few simple design best practices can ensure a pack level explosion is prevented even if a single cell undergoes TR. To understand this, we first need to understand how TR occurs in a battery pack. Simply put, a battery pack consists of a number of individual cells stacked next to and connected to each other. This assembly of cells is called a module and several modules together make a pack. Whenever a battery is in operation (whether charging or discharging), each of these cells generate heat and increase the temperature of the cells. Since they are in close proximity to each other, this heat is transferred to neighbouring cells.
NMC 18650 Cell Temperature: ARC Test Data
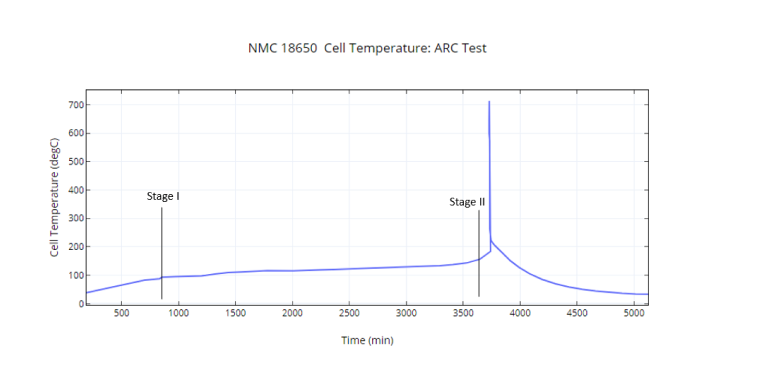
Figure 1. Experimental data denoting the onset of Thermal Runaway for an NMC 18650 cell. Stage 1 indicates the onset of electrode decomposition and Stage 2, the onset of electrolyte decomposition
Traditional Cooling Approaches can be a Double-edged Sword
Batteries are highly sensitive to temperatures and high temperatures (anything above ~ 40oC) can significantly affect the life and the performance of a battery. Battery designers therefore use several strategies to reduce temperature in packs by ensuring that the heat generated in one cell quickly propagates to the neighbouring cell to help dissipate the heat quickly. This can be achieved though various approaches such as active cooling, where a coolant is run across, above and below cells, or using thermal pads between cells to propagate heat, or placing fans around the pack to distribute the heat evenly. All of this works very well during normal operation.
The problem starts when one of the cells reaches a very high temperature for some reason (usually due to some abuse): also called the onset temperature. Beyond this temperature (usually around 80-100oC), the temperature of this cell start increasing extremely rapidly (to about 140oC in a few seconds, see Fig 1).
The traditional methods of cooling fail here and in fact, promote thermal runaway because they spread the heat rapidly. When a cell experiencing TR passed on the heat to the neighbouring cells, it causes them to start experiencing TR too. These reactions are also accompanied by a release of combustible gases at very high speeds. With a majority of the cells in the pack at uncontrollably high temperature, it leads to an explosion that you see on those viral videos.
Predicting Thermal Runaway is Hard
The reason it is difficult to predict this phenomenon for researchers is because of the short timescales of the entire process: the entire process from the onset of TR (~ 100oC) to reaching the maximum temperature (~ 300oC), accompanied by the high velocity release of combustible gases can happen in less than a second and the propagation through the pack takes less than 5s. The speed and amount of energy release makes observations exceedingly difficult and expensive. The reason the phenomenon is misunderstood in the public domain is because of the fear associated with the videos of vehicles suddenly engulfed in flames within a matter of seconds on smoke coming out of the pack. The assumption is that batteries are unpredictable and every time there is TR in a cell, the pack is bound to explode. This is far from the truth.
Adopting Design Best Practices
In reality, some simple design best practices can ensure the risk of TR is mitigated. Research has shown that even if a cell undergoes TR, with good design the rest of the cells can be protected, and an explosion can be averted. The same research has also shown that even simple steps like increasing the thickness of the aluminum casing, or increasing the distance between cells can slow down TR propagation thereby eliminating explosion. Other strategies such as inserting insulating pads can prevent TR propagation between cells.
However, the questions are, what is the minimum thickness of the casing and the minimum spacing between cells to ensure TR propagation is slowed to the extent an explosion is prevented? Which insulating material can we use to ensure TR propagation is mitigated yet thermal management during regular operation is not impacted, since the requirements of thermal management during regular operations are exactly the opposite of those of TR mitigation.
Cell Temperatures: NMC Cell
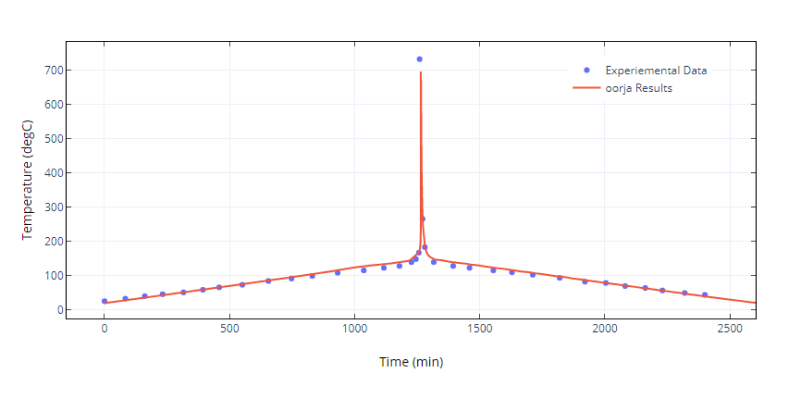
Figure 2. Validation with experimental results performed on an 18650 NMC cell
At oorja, we have developed an application which allows our users to predict this phenomenon quickly and accurately, thereby enabling them to optimize battery design and ensure TR mitigation.
Using our patent pending approach of using a combination of physics based and experimental data to perform training and then run predictions for different design configurations, users can optimize their pack design and cooling strategies and ensure the risk of explosion is minimized. Oorja also provides the ability to perform necessary experiments to ensure users don’t have to worry about performing the experiments themselves.
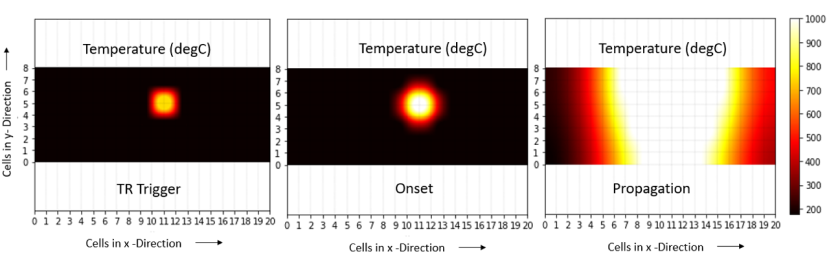
Figure 3. Thermal Runaway Propagation at the pack level
Validation of oorja results with experimental results in cells can be seen in Fig 2 which shows how the temperature in a cell shoots up during the TR process. Further, Fig 3 shows how the temperature propagates through the pack while Fig 4 shows the temperature rise in each of the cells of a pack with time.
Cell Temperatures
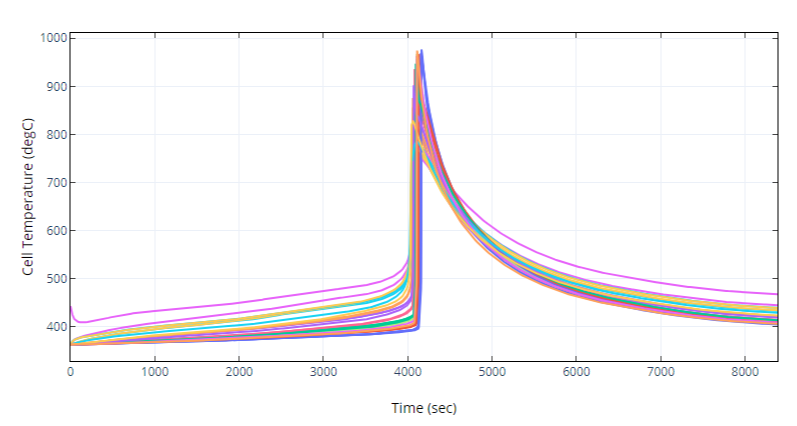
Figure 4. oorja results for temperature rise in all cells of a pack undergoing TR.
Contact us if you need to learn more about our approach or to use our application to mitigate the risk of TR.
Latest Blog Posts
Hybrid Modeling: Bridging Physics and Machine Learning
Hybrid Modeling: Bridging Physics and Machine Learning Get Started Contact...
Read MoreAccelerated Degradation Predictions using Hybrid (Physics+ML) Simulations
Accelerated Degradation Predictions using Hybrid (Physics+ML) Simulations Get Started Contact...
Read More


